HDX-MS: See what sequence alone cannot reveal
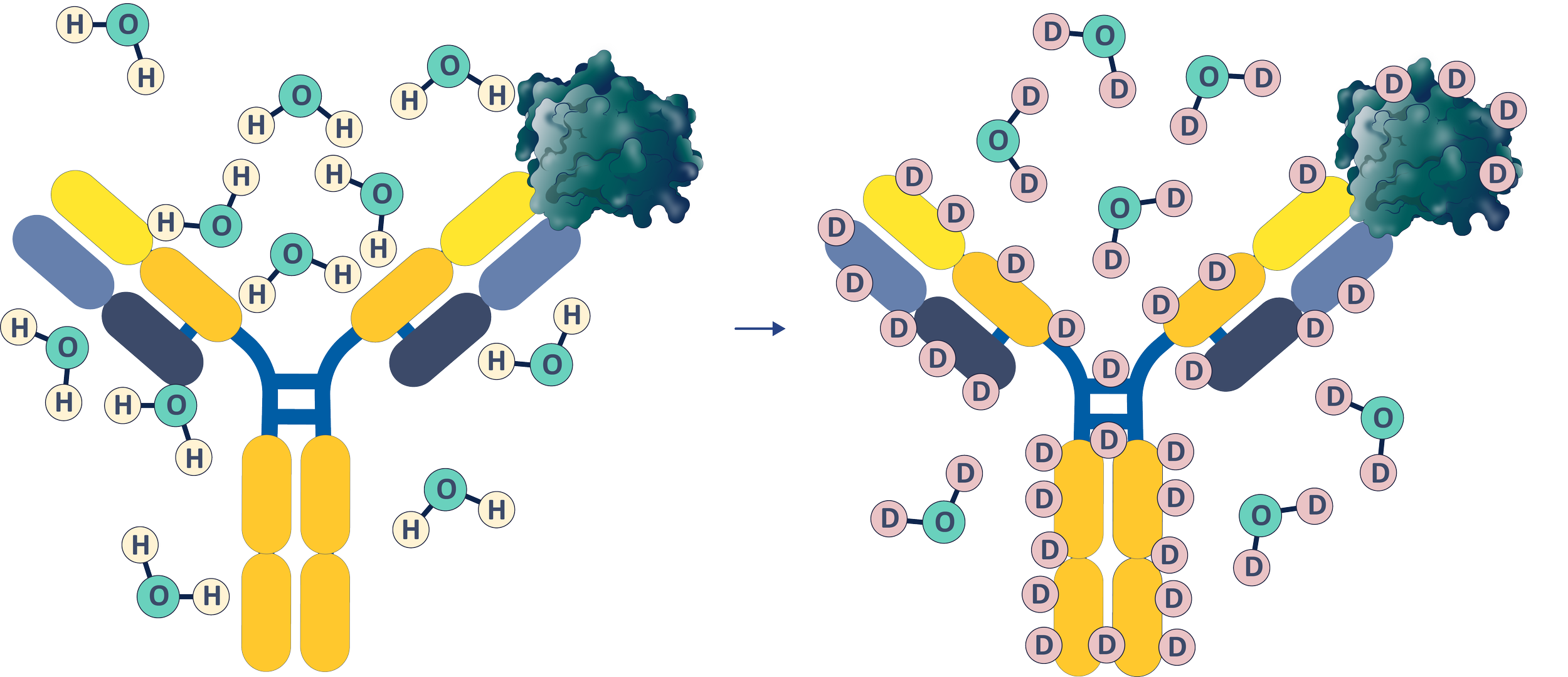
Hydrogen-deuterium exchange mass spectrometry delivers residue-level insight into protein conformation, dynamics, and interaction surfaces — essential data for biosimilarity assessments, structural elucidation, and comparability studies.
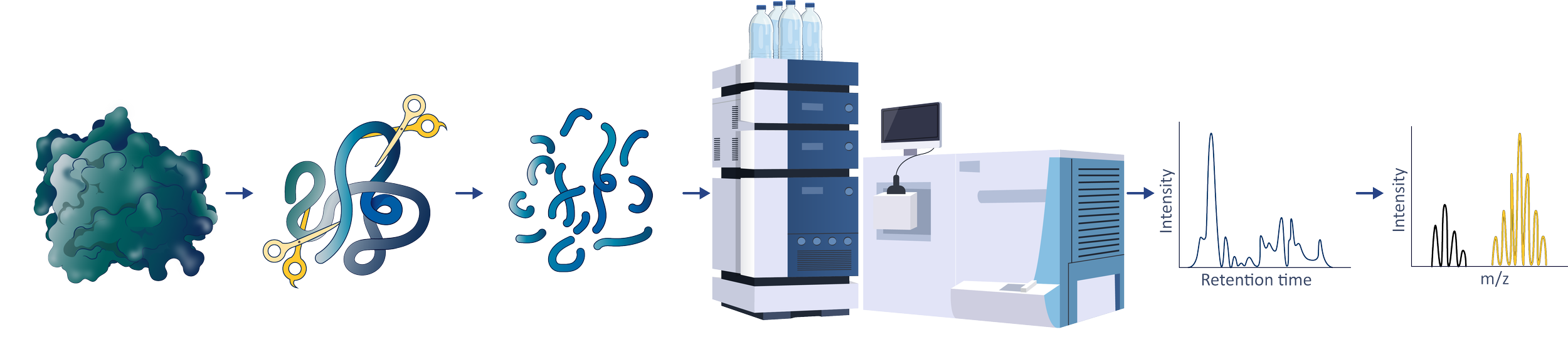
The HDX-MS Workflow
Labeling
& Digest
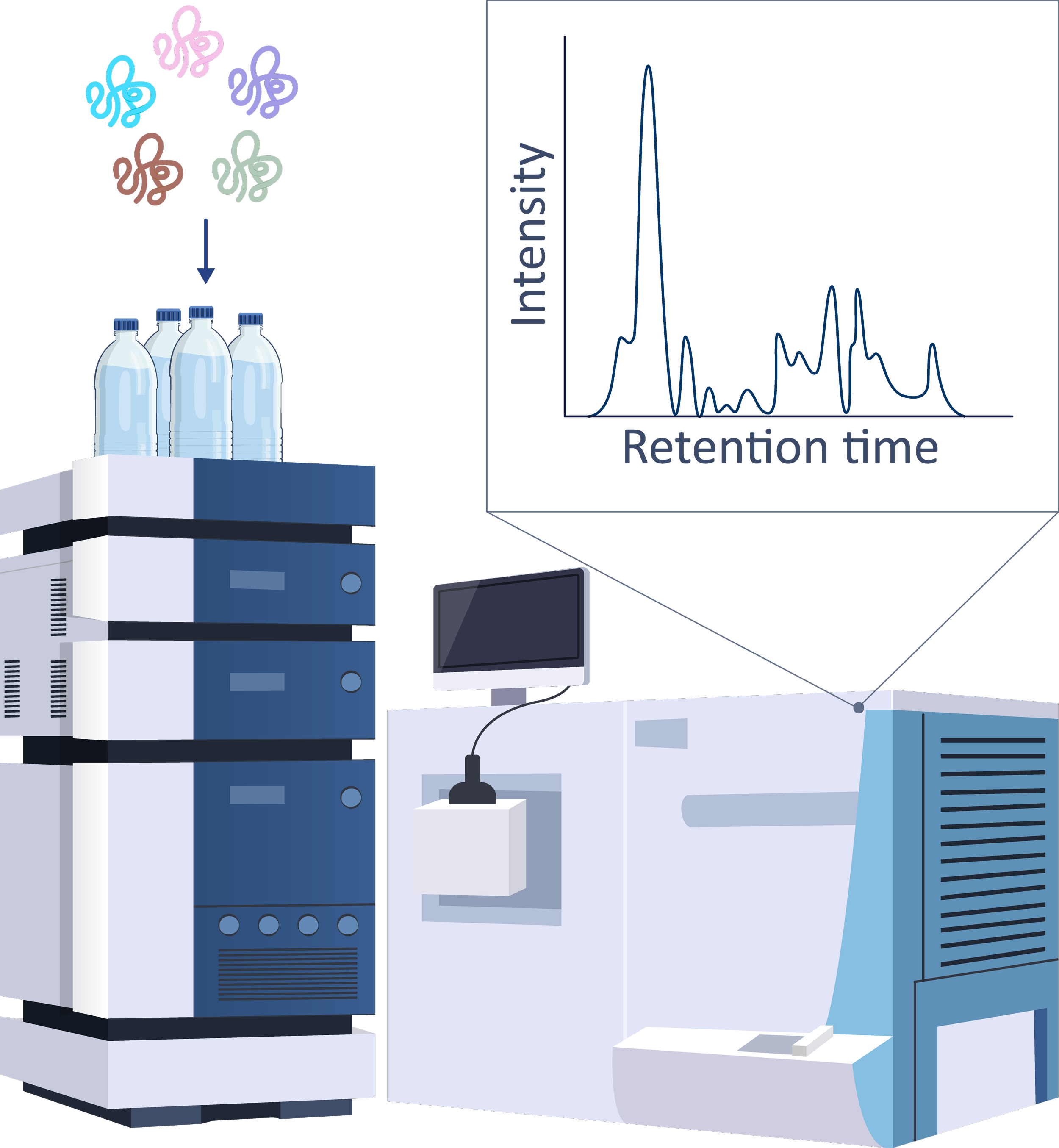
Analysis
Processing
Interpretation
Epitope Mapping
Precisely localize antibody-antigen binding interfaces at peptide-level resolution. Define conformational and linear epitopes to inform rational design.
Conformational Stability
Monitor higher-order structure changes across formulations, stress conditions, and manufacturing processes with quantitative deuterium uptake profiles.
Biosimilarity Assessment
Generate the higher-order structure comparability data required for biosimilar regulatory submissions with validated, reproducible methods.
Binding Interfaces
Characterize protein-protein, protein-ligand, and multi-component binding surfaces to support mechanism-of-action studies and formulation development.
Comparability Studies
Detect higher-order structure changes across manufacturing process changes, site transfers, and lot-to-lot variation with quantitative HDX-MS comparisons.
Forced Degradation
Evaluate the structural impact of thermal, oxidative, and pH stress on higher-order structure to identify degradation-sensitive regions and inform stability programs.
Formulation Development
Screen formulation candidates by monitoring HOS changes across buffer, excipient, and concentration conditions to select optimal formulations early.
Mechanism of Action
Map conformational changes upon ligand binding, receptor engagement, or allosteric modulation to support MoA characterization and rational drug design.